
Introduction
The article “Interpreting and Reporting Clinical Trials with Results of Borderline Significance” by Allan Hackshaw and Amy Kirkwood, published in the BMJ, addresses a critical issue in clinical research: the interpretation of borderline statistical significance, particularly concerning p-values around the conventional threshold of 0.05. This blog aims to elucidate the key points of the article for statisticians, clinicians, and clinical researchers, emphasizing the implications of p-values and confidence intervals in clinical trial reporting.

The Problem with P-ValuesThe authors argue that a p-value just above or below 0.05 can lead to misleading conclusions about treatment efficacy. For instance, a relative risk of 0.75 with a p-value of 0.048 suggests a significant effect, while a similar relative risk with a p-value of 0.051 might be dismissed as having no effect. This binary interpretation fails to capture the nuances of statistical evidence and can lead to erroneous clinical decisions.Confidence Intervals as a SolutionThe article highlights that confidence intervals (CIs) provide more informative insights than p-values alone. For example, in the EICESS-92 trial on Ewing’s sarcoma treatment, the observed hazard ratio was 0.83 with a 95% CI of 0.65 to 1.05 (p=0.12). Although this result does not meet the conventional significance threshold, the CI indicates that there is still a substantial probability that the treatment may be beneficial.
Hazard Ratio Interpretation
Figure: Interpretation of Hazard Ratio with Confidence Interval This graph illustrates that while the CI includes the value of 1 (no effect), most of its range lies below it, indicating a higher likelihood of a beneficial treatment effect.

Variability in ConclusionsThe authors conducted an analysis of abstracts from major medical journals and found significant inconsistencies in how borderline results were reported. For instance, some studies with similar p-values and effect sizes reached different conclusions about efficacy. This inconsistency can confuse clinicians and researchers trying to interpret trial results.Examples from LiteratureThe article provides examples where studies reported no effect despite having borderline significant results. For instance:
These examples highlight how language can shape interpretation and potentially mislead stakeholders regarding treatment efficacy
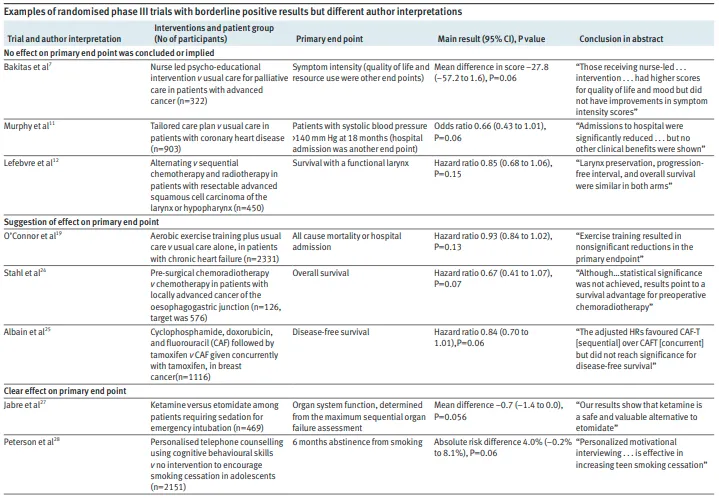
Standardizing Reporting PracticesTo address these issues, Hackshaw and Kirkwood recommend standardizing how borderline results are reported across journals and studies. They suggest that authors should:
Utilizing Surrogate EndpointsAnother recommendation is to consider using validated surrogate markers as primary endpoints in trials where feasible. This approach could increase event rates and improve statistical power, reducing the occurrence of borderline results.
The article by Hackshaw and Kirkwood serves as a vital reminder for statisticians, clinicians, and researchers about the complexities surrounding statistical significance in clinical trials. By emphasizing confidence intervals over arbitrary p-value cutoffs and advocating for clearer reporting standards, we can enhance the quality and reliability of clinical research findings.In summary, understanding and interpreting borderline significance is crucial for informed decision-making in clinical practice, ensuring that potentially beneficial interventions are not overlooked due to rigid adherence to outdated statistical conventions.